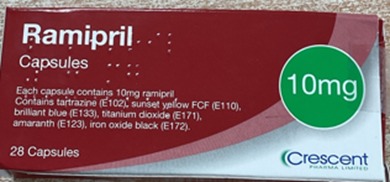
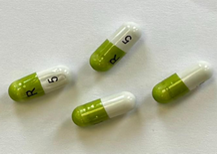
Some cartons of Ramipril 10mg Capsules, manufactured by Crescent Pharma Limited, may contain blister strips of Ramipril 5 mg Capsule.
All packs from the impacted batch of Ramipril 10 mg Capsule, (Batch Number GR174091) are being recalled as a precautionary measure.
If you were prescribed Ramipril 10 mg Capsules and have received the impacted product batch (Batch Number GR174091) please check that the carton contains the correct medication. The batch number and expiry date information can be found on outer carton.
If the carton contains blister strips that are labelled and contain Ramipril 5 mg capsule, contact your pharmacy in the first instance. If the carton contains blister strips that are labelled and contain Ramipril 10 mg Capsules, you do not need to take further action.
If you are unsure or have any questions, please seek advice from your pharmacy or the dispensary team.
Please take the leaflet that came with your medicine and any remaining capsules with you to your pharmacy or GP practice.
Both strengths are used to treat high blood pressure, heart failure and kidney disease. Any possible impact of a lower dose of ramipril is expected to be gradual rather than immediate or life threatening.
For reference, the description of the products are as follows:
Ramipril 10 mg Capsules: Capsules are light grey and dark green capsules, marked with “R” on the cap and “10” on the body.
Ramipril 5mg Capsule: Capsules are light grey and green gelatin capsules, marked with “R” on the cap and “5” on the body.